CELL CULTURE & TISSUE ENGINEERING
Applications
Conventional cell culture systems do not reproduce the 3D complexity or physiological conditions of living tissue. They limit control of the microenvironment and the homogeneity of nutrient and oxygen supply. 3D microfabrication, such as our microFAB-3D system, makes it possible to design structures with fine architecture, controlled porosity, and permeability to exchanges to more closely match in vivo conditions.
microFAB-3D and Smart Print UV can be used with biocompatible materials, which have low cytotoxicity after polymerization. Printing can be performed directly on standard substrates (glass slides, functionalized surfaces, well plates…), facilitating integration into conventional biological workflows.
To help with cell culture, heaters and sensors can also be made using Smart Print UV, for a more precise heat generation.
Both machines can also be used together to design systems for dynamic cell culture, integrating, for example, microfluidic networks to simulate physiological flows or organ-on-chip devices.
Highlight
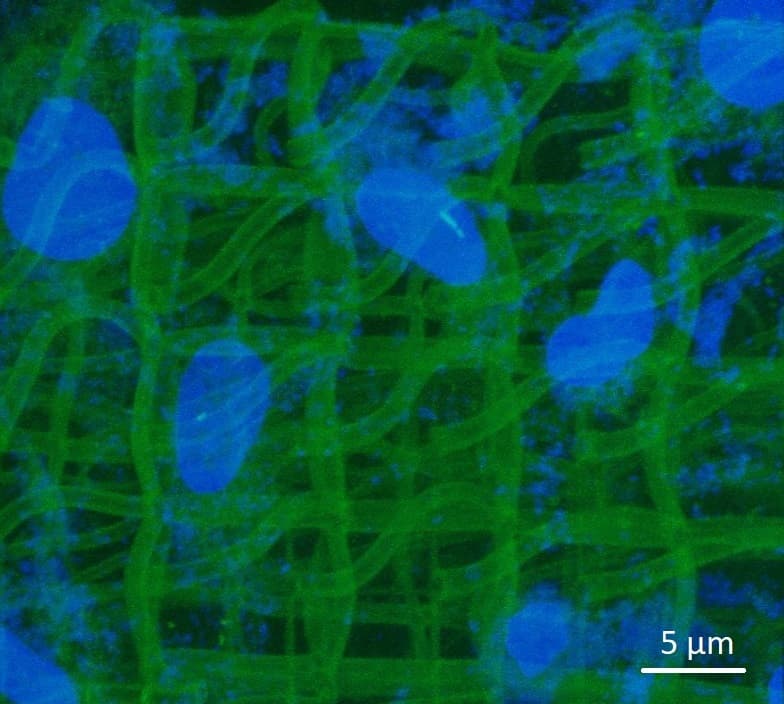
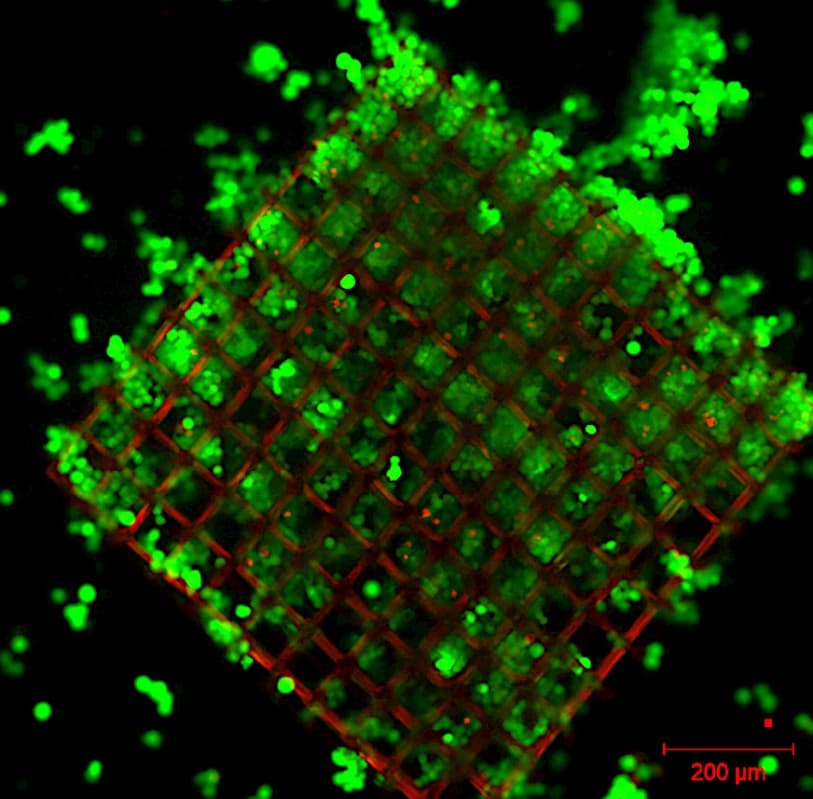
Gelatin and collagen gels are good candidates for the development of cell culture media. Collagen, by its self-association properties, forms the weft of most tissues. In a biomimetic approach, collagen combined with gelatin therefore seems an interesting candidate to reconstitute the dermis. A three-dimensional (3D) structuring of gels is presented: formation of cubic, alveolar or more open structures such as stacks of hollow cubes.
This 3D structuring is made possible by the use of a laser microprinting system marketed by Microlight3D. With this system we spatially control the cross-linking of proteins, a phenomenon that occurs at the focal point of the micro-laser.
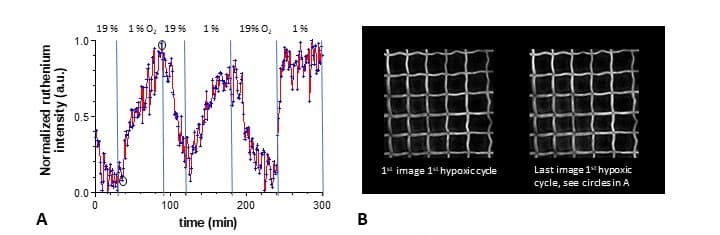
For this process it is necessary to add to the starting medium a photoinitiator, in our case a ruthenium complex. This initiator, which allows the solidification of the medium at the desired locations, also has interesting optical properties with oxygen. Its photoluminescence increases when the amount of oxygen in its environment decreases and decreases when the amount of oxygen present nearby increases. Thus, by following the intensity of ruthenium luminescence on the 3D structures that we have manufactured and colonized by living cells we have shown that we are able to highlight locally (at cell size scales) the activity of these cells when they migrate on the gel structures or when they divide on these same 3D structures. Indeed in this case they consume oxygen which can be followed by the decrease in the fluorescence intensity of ruthenium present in 3D structures.
Olivier Stephan is a teacher-researcher in the University Grenoble Alps (UGA).